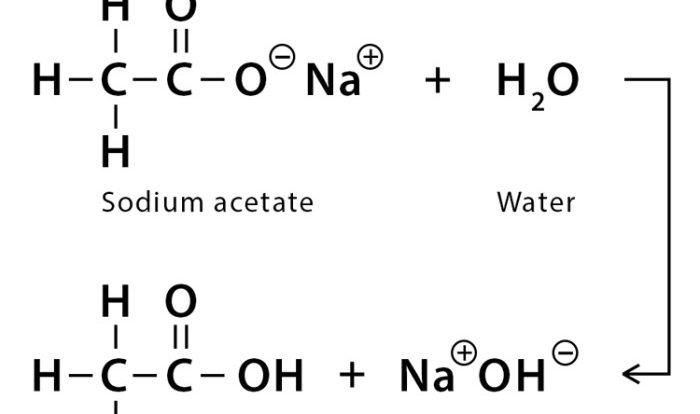
In aqueous solution classify these compounds as strong – In aqueous solution, classifying compounds as strong involves understanding their behavior and properties in water. Strong acids and bases exhibit distinct characteristics that impact their reactivity and applications. This exploration delves into the factors that determine the strength of these compounds, their neutralization reactions, and their practical significance in various fields.
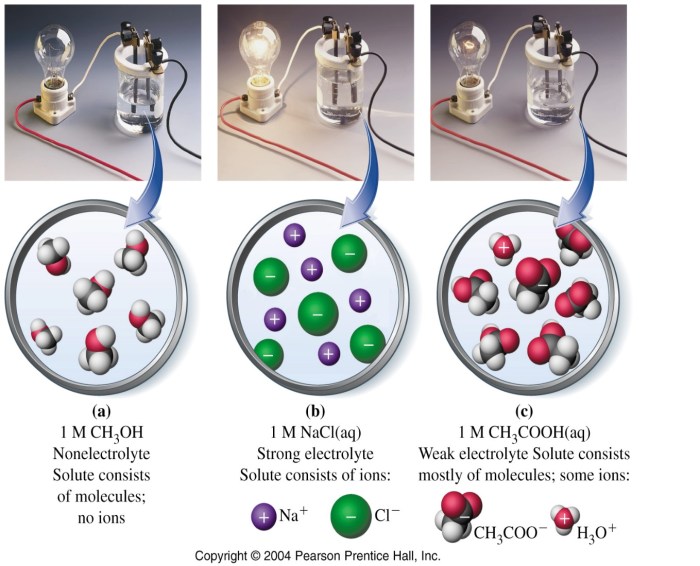
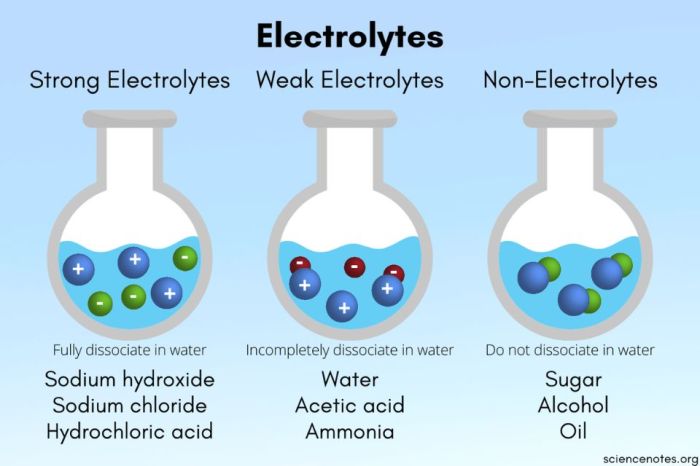
Strong acids, such as hydrochloric acid and sulfuric acid, completely dissociate in water, releasing hydrogen ions (H+) and contributing to a low pH. Strong bases, like sodium hydroxide and potassium hydroxide, release hydroxide ions (OH-) upon dissolution, resulting in a high pH.
The strength of an acid or base is influenced by factors such as bond strength, hydration energy, and the stability of the conjugate base or acid.
Strong Acids

Strong acids are chemical compounds that completely dissociate in aqueous solutions, releasing hydrogen ions (H +) and anions. They have a pH of less than 7 and are highly corrosive.
Common strong acids include:
- Hydrochloric acid (HCl)
- Sulfuric acid (H 2SO 4)
- Nitric acid (HNO 3)
- Hydroiodic acid (HI)
- Hydrobromic acid (HBr)
The strength of an acid is determined by its dissociation constant (K a). The higher the K a, the stronger the acid.
Strong Bases

Strong bases are chemical compounds that completely dissociate in aqueous solutions, releasing hydroxide ions (OH –) and cations. They have a pH greater than 7 and are highly caustic.
Common strong bases include:
- Sodium hydroxide (NaOH)
- Potassium hydroxide (KOH)
- Calcium hydroxide (Ca(OH) 2)
- Lithium hydroxide (LiOH)
- Barium hydroxide (Ba(OH) 2)
The strength of a base is determined by its dissociation constant (K b). The higher the K b, the stronger the base.
Neutralization Reactions: In Aqueous Solution Classify These Compounds As Strong
Neutralization reactions occur when a strong acid and a strong base react in aqueous solution. The products of a neutralization reaction are a salt and water.
For example, the reaction between hydrochloric acid (HCl) and sodium hydroxide (NaOH) produces sodium chloride (NaCl) and water (H 2O):
HCl + NaOH → NaCl + H2O
The pH of a neutralization reaction is 7, indicating a neutral solution.
Applications of Strong Acids and Bases
Strong acids and bases have numerous applications in various industries:
Strong Acids, In aqueous solution classify these compounds as strong
- Pickling and cleaning metals
- Manufacturing fertilizers
- Producing dyes and pigments
- Electroplating
- Oil refining
Strong Bases
- Manufacturing soap and detergents
- Producing paper and textiles
- Treating wastewater
- Neutralizing acids
- Making food additives
Strong acids and bases are essential in everyday life, from cleaning products to industrial processes.
Expert Answers
What is the difference between a strong acid and a weak acid?
Strong acids completely dissociate in water, releasing all their hydrogen ions, while weak acids only partially dissociate.
How can I determine the strength of a base?
The strength of a base is determined by its ability to accept protons (H+ ions), which can be measured using the pKb value.
What is the pH of a neutral solution?
A neutral solution has a pH of 7, indicating equal concentrations of hydrogen ions (H+) and hydroxide ions (OH-).